|
We reasoned that overexpression might allow for increased availability of regenerative machinery within the axon, leading to better regeneration after injury. Our initial studies found that Protrudin mRNA is expressed at low levels in CNS neurons, but at higher levels in regenerating PNS neurons, and the protein is present in immature regenerative CNS axons but is depleted from axons with maturity as regeneration is lost. Our hypothesis was that the expression of an active form of Protrudin would enable regeneration of CNS axons via a combination of these interactions and would be a powerful tool for understanding the mechanisms of axon regeneration. Protrudin therefore links a number of cellular components associated with axonal growth. Specific phosphoinositides are required at the growth cone during rapid axon growth and for axon regeneration 26. Protrudin also has a FYVE domain that binds to phosphoinositides enabling interaction with endosomes and the surface membrane 25. In addition to its localization at ER tubules, Protrudin also regulates ER distribution and network formation 23. This interaction is involved in its effects on protrusion outgrowth 23, 24, 25. Protrudin localizes to the ER through two transmembrane domains and a hairpin loop and interacts with VAP proteins at ER-membrane contact sites through an FFAT domain. 
This is relevant to CNS axon repair because increased Rab11 transport into CNS axons in vitro increases their regenerative ability 19. Through its Rab11 and kinesin-1 (KIF5) binding sites, Protrudin can enable the anterograde transport of Rab11-positive recycling endosomes, leading to their and their cargo’s accumulation at protrusion tips 21, 22. 
First, overexpression of Protrudin causes protrusion formation in non-neuronal cell lines, and promotes neurite outgrowth in neuronal cells 21 second, Protrudin is a scaffolding molecule which possesses interaction sites for key axon growth-related molecules and structures 21. Protrudin is an integral endoplasmic reticulum (ER) membrane protein that has two properties which make it a candidate for enabling axon regeneration. This study focuses on the adaptor molecule Protrudin as a tool for investigating and enhancing axon growth and regeneration in the adult CNS. However, the precise machinery required to reconstitute and extend an injured axon is not completely understood, and repairing the injured CNS remains a challenging objective 20. Studies aimed at enhancing CNS regeneration have identified transcriptional and epigenetic programs 6, 7, signalling pathways 8, 9, 10, the cytoskeleton 11, 12, 13, 14 and axon transport 15, 16, 17, 18, 19 as important factors governing regenerative ability. In addition to the non-permissive extracellular environment after injury, intrinsic neuronal factors also play an important role in the regenerative failure observed in mature CNS neurons 4, 5. In contrast, adult CNS neurons lose their regenerative ability with maturation 3, meaning that axonal injury or disease has life-altering consequences and that there is little chance of recovery. These results demonstrate that Protrudin promotes regeneration by functioning as a scaffold to link axonal organelles, motors and membranes, establishing important roles for these cellular components in mediating regeneration in the adult central nervous system.Īxons of immature central nervous system (CNS) and adult peripheral nervous system (PNS) neurons readily regenerate after injury 1, 2. Protrudin overexpression facilitated the accumulation of endoplasmic reticulum, integrins and Rab11 endosomes in the distal axon, whilst removing Protrudin’s endoplasmic reticulum localization, kinesin-binding or phosphoinositide-binding properties abrogated the regenerative effects. Elevated Protrudin expression enabled robust central nervous system regeneration both in vitro in primary cortical neurons and in vivo in the injured adult optic nerve. 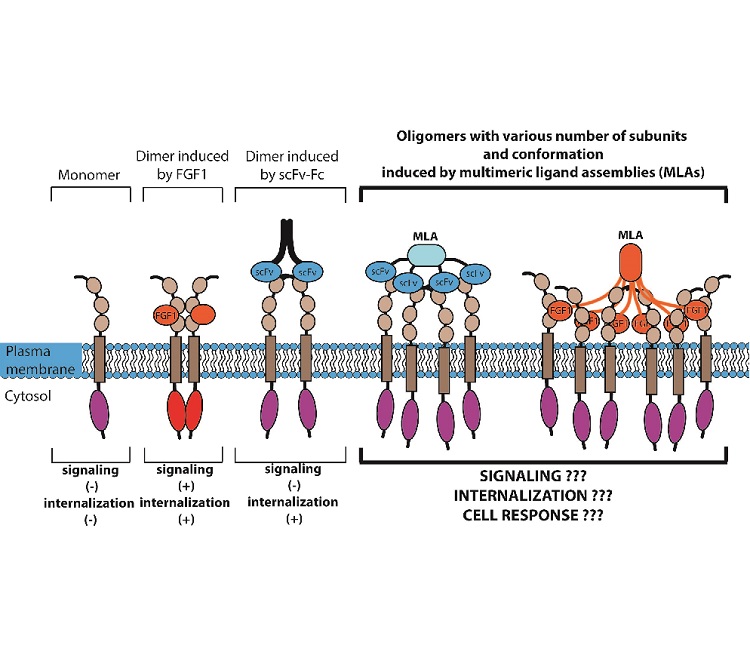
We achieved this by expressing the adaptor molecule Protrudin which is normally found at low levels in non-regenerative neurons. One approach to enhancing regeneration is to increase the axonal supply of growth molecules and organelles. Adult mammalian central nervous system axons have intrinsically poor regenerative capacity, so axonal injury has permanent consequences.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
